TNO's Digital Biomarker Lab
See more, treat better
The TNO Digital Biomarker Lab reveals meaningful health patterns from daily life through patient-centric digital biomarkers. We turn digital measures into trusted evidence for clinical research and healthcare decision-making.
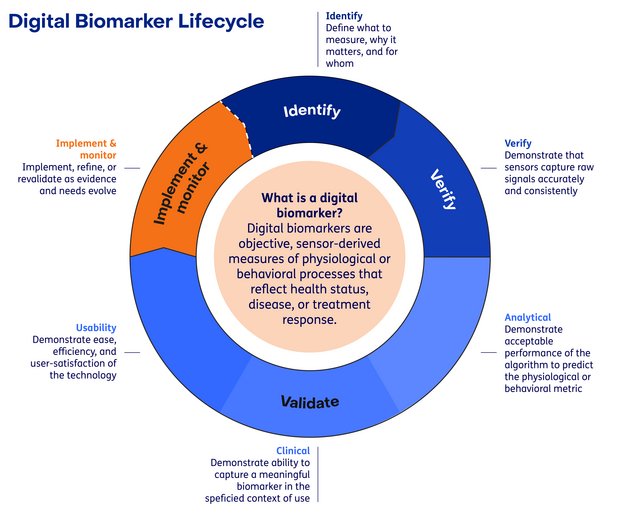
Why Digital Biomarkers?
Across everyday life, care pathways and clinical studies, people generate rich signals about health, behaviour and treatment response. Yet these signals are rarely captured, validated or translated into meaningful evidence that patients, clinicians and decision-makers can rely on.
Traditional clinical measures are episodic and often burdensome, capturing only a limited snapshot of a person’s health. Important information about how people function, respond to treatment and recover between visits can be missed, despite its relevance for care and evaluation.
Digital biomarkers offer a promising solution. By collecting objective signals more continuously in daily life, they can complement traditional measures and provide a richer picture of health and treatment response outside the clinic. Digital biomarkers are derived from digital measures , which are raw sensor -based outputs without intrinsic clinical meaning, and are transformed into digital biomarkers once they are interpreted and validated for their clinical relevance.
To ensure this transformation and that digital biomarkers are fit for purpose, their development and evaluation can be structured using established frameworks such as the Digital Medicine Society’s ( DiMe) V3+ framework for verification and validation, together with the Digital Measures That Matter framework, which anchors measurement selection in patient -meaningful aspects of health.
Services: digital biomarker discovery, validation and remote data collection
At the TNO Digital Biomarker Lab, we use a structured pathway to develop digital biomarkers that are trustworthy, interpretable, and meaningful in daily life, so they can support decisions in clinical research and healthcare.
We take a rigorous, independent, and patient-centric approach. We start with the question that matters most: what do patients and clinicians need to understand, monitor, or improve? From there, we define the intended context of use and build the evidence package required for adoption, scientifically, operationally, and, where relevant, regulatory-aligned.
We support partners across key stages of the digital biomarker lifecycle. By combining discovery, validation, patient engagement and data infrastructure, we enable clients and partners to build digital biomarkers that are robust, interpretable and ready to be used, whether in clinical research, healthcare practice or real-world settings.
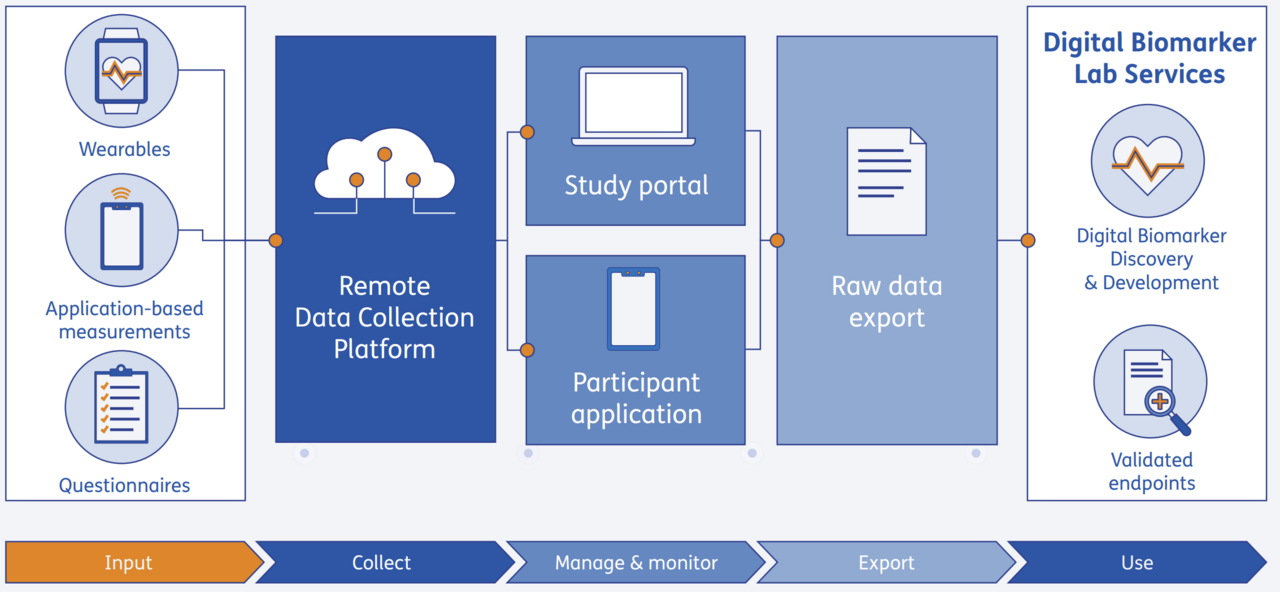
Remote Data Collection Infrastructure
device-agnostic platform for secure digital health studies
Remote studies often involve multiple devices and data streams, but many platforms are tied to a single vendor ecosystem. This limits device choice and makes it harder to combine wearable data, app-based measures, and questionnaires into one study dataset. At the same time, certified infrastructure for sensitive health data is scarce, so teams spend time building or setting up their own infrastructure with compliant workflows before a study can begin.
TNO has built a platform to overcome these challenges: the platform provides secure, ISO- and NEN-certified infrastructure that is GDPR compliant and suitable for sensitive health data, designed for clinical and research environments, with a flexible model that supports both feasibility pilots and larger deployments. The platform can also connect to EHR (Electronic Health Record) systems in hospitals, enabling seamless integration with existing clinical workflows.
Because the integration approach is flexible (including 'Bring Your Own Device'), the platform can be configured to connect with a wide range of consumer-grade wearables and apps, as well as medical-grade apps and novel/customer-specific devices - so you are not locked into one device ecosystem.
Study teams can configure data capture and monitor data quality, participant progress, and logistics in a study portal. Participants use a dedicated app for remote onboarding and device pairing, and can receive reminders and personal data visualisation (real-time feedback can be enabled or switched off where needed). Study execution support can also be added to help keep studies running smoothly and data quality high. Importantly, the platform provides full access to raw data, enabling transparent, research-grade analysis and publication-ready results.
Contact information
Interested in more information about the work and projects that we are doing? Contact us at: meike.vandeneijnden@tno.nl
